|
4/11/2024 0 Comments Define absolute entropy
Here we further explore the nature of this state function and define it mathematically. 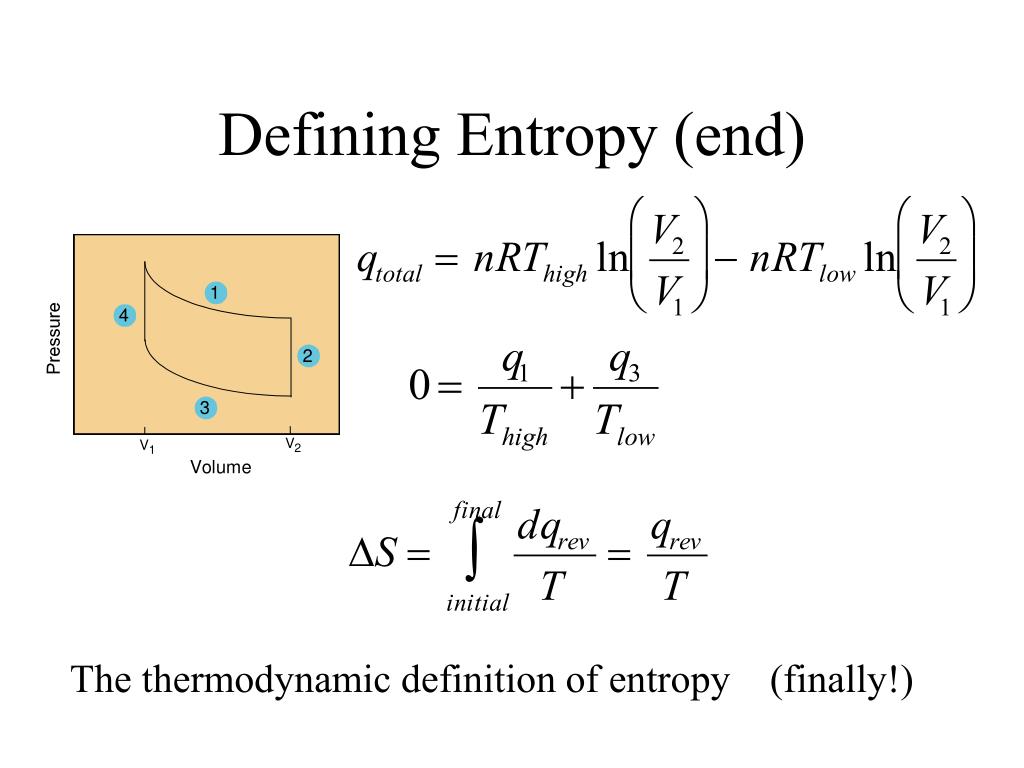
In Chapter 13, we introduced the concept of entropy in relation to solution formation. To help explain why these phenomena proceed spontaneously in only one direction requires an additional state function called entropy (S), a thermodynamic property of all substances that is proportional to their degree of "disorder". Moreover, the molecules of a gas remain evenly distributed throughout the entire volume of a glass bulb and never spontaneously assemble in only one portion of the available volume. However, the absolute value of the entropy of a physical system and the more general creative meaning and value of entropy were not clearly defined and explained by Clausius, which made the Clausius entropy a little mysterious and speculative. For example, after a cube of sugar has dissolved in a glass of water so that the sucrose molecules are uniformly dispersed in a dilute solution, they never spontaneously come back together in solution to form a sugar cube. For a full video: see Thus enthalpy is not the only factor that determines whether a process is spontaneous. Standard entropy of pure substance (So) : Entropy of a pure substance in its standard state that is pressure 1 bar and the temperature 298 K, is called the. When water is placed on a block of wood under the flask, the highly endothermic reaction that takes place in the flask freezes water that has been placed under the beaker, so the flask becomes frozen to the wood. The reaction of barium hydroxide with ammonium thiocyanate is spontaneous but highly endothermic, so water, one product of the reaction, quickly freezes into slush. The meaning of ENTROPY is a measure of the unavailable energy in a closed thermodynamic system that is also usually considered to be a measure of the system's disorder, that is a property of the system's state, and that varies directly with any reversible change in heat in the system and inversely with the temperature of the system broadly : the degree of disorder or uncertainty in a system. The third Law of Thermodynamics states that the entropy of a pure substance in a perfect crystalline form is 0 J molK J m o l K at 0 K: S 0 K 0 S 0 K 0. Melting of a solid and vaporization of a liquid correspond to sizeable increases in the number of microstates available to accept thermal energy, so as these processes occur, energy will flow into a system, filling these new microstates to the extent required to maintain a constant temperature (the freezing or boiling point) these inflows of thermal energy correspond to the heats of fusion and vaporization.\): An Endothermic Reaction. We can use this to define a natural zero, giving entropy an absolute scale. As far as a formula for entropy, well there isn’t just one. where Q3 Q 3 is the amount of heat taken from the hotter reservoir and Q2 Q 2 is the. We have already seen that the efficiency of a Carnot cycle is given by. Now we can show this just from the laws of thermodynamics. For any chemical reaction, the standard entropy change is the sum of the standard molar entropies of the products minus the sum of the standard molar entropies of the reactants. 1 mol of NH 3 (g) or 1 mol of He (g), both at 25☌. Although we have used the notion of absolute temperature, it was not proven. The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. 
Predict which substance in each pair has the higher entropy and justify your answer. Since entropy is primarily dealing with energy, it’s intrinsically a thermodynamic property (there isn’t a non-thermodynamic entropy). The magnitude of the increase is greater than the magnitude of the decrease, so the overall entropy change for the formation of an NaCl solution is positive. The enthalpies associated with any phase changes the substance may undergo within the temperature range of interest. First it’s helpful to properly define entropy, which is a measurement of how dispersed matter and energy are in a certain region at a particular temperature.Two kinds of experimental measurements are needed: The absolute entropy of a substance at any temperature above 0 K must be determined by calculating the increments of heat \(q\) required to bring the substance from 0 K to the temperature of interest, and then summing the ratios \(q/T\). The Third Law Lets us Calculate Absolute Entropies 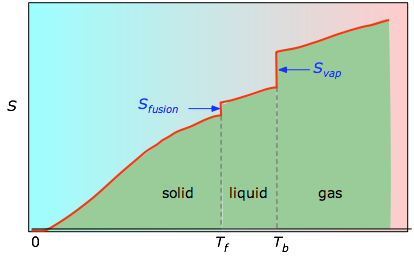
The entropy again increases steadily with increasing temperature until the boiling point is reached, where it jumps suddenly as the liquid undergoes a phase change to a highly disordered gas (ΔS vap). Absolute entropy increases steadily with increasing temperature until the melting point is reached, where it jumps suddenly as the substance undergoes a phase change from a highly ordered solid to a disordered liquid (ΔS fus). \): A Generalized Plot of Entropy versus Temperature for a Single Substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
